Nitrogen-Fixing Bacteria in Compost Extracts: DNA analysis case study
- Jan 26
- 6 min read
Updated: Jan 27

At a Glance: Nitrogen-Fixing Bacteria in Compost Extracts
DNA sequencing shows that nitrogen-associated rhizobacteria (Rhizobiales) occur at similar relative abundance (~5%) across well-made compost extracts, regardless of compost type.
Compost DNA percent composition is not a measure of biomass and should be interpreted alongside microscopy or CFU data.
Microbial diversity matters more than the presence of any single organism.
Bacterial biomass was highest in the Early-Successional compost blend, making it the best choice when maximizing rhizobacterial abundance is the primary goal.
Compost selection should consider biomass, diversity, handling, certifications, and cost, not just taxonomic presence.
Key takeaway: Most high-quality composts contain nitrogen-fixing bacteria — the practical difference lies in how much biology is present and how well it matches the intended application.
A friend recently asked me whether my compost extracts or compost teas contained rhizobacteria — nitrogen-fixing bacteria associated with plant roots. I had to dig into my DNA amplicon sequencing data to answer confidently.
To simplify the question, I focused my analysis on my three compost extracts, rather than compost teas. Microbial community structure and diversity can shift significantly when a compost extract is brewed into a compost tea, and I wanted to examine the biology of the composts themselves as inoculant sources.
Compost Extract Recipes and Successional Design
In early 2025, I designed three compost extract (and corresponding compost tea) recipes based on plant and microbial succession theory. Each recipe was intended to align with a different plant successional stage and was built around a distinct compost biology:
Early-Successional ExtractMade from a bacterial-dominant compost blend.
Mid-Successional ExtractMade from a highly diverse compost with a fungal-to-bacterial (F:B) ratio of approximately 0.5–1.
Late-Successional ExtractMade from a fungal-dominant compost (F:B > 1).
While these recipes are blends, their dominant compost inputs differ:
The Early-Successional compost is primarily a commercial, bacterial-dominant compost.
The Mid-Successional compost is my home-made thermal aerobic compost, produced according to Dr. Elaine Ingham’s standards for BioComplete™ compost.
The Late-Successional compost is primarily a home-made Johnson-Su compost.
At the time of DNA testing, my Johnson-Su compost was still partially frozen at the core, so I supplemented the recipe with a small amount of my farm-made, 1.5-year-old “Alpha” compost, which was very high in carbon and fungal biomass.
Interpreting DNA Sequencing Data: Strengths and Limits
DNA amplicon sequencing results are often overwhelming — not just for growers, but for researchers and academics as well. Thousands of bacterial taxa, hundreds of fungi, and dozens of eukaryotes (including protozoa, nematodes, and other microfauna) can be detected in a single sample.
To make sense of the data, I initially sorted taxa by number of sequence hits to identify the most abundant groups, then searched the literature to understand their known or proposed functions.
What I found was humbling but unsurprising:most detected microbes have little to no functional information available at the genus or species level.
While we understand a great deal about a small subset of organisms that can be cultured and studied in the lab, the vast majority of soil microbes — often estimated at >99% — are unculturable and remain poorly characterized. In many ways, we still know more about outer space than about the first six inches of soil.
Why I Focus on Diversity
In ecological systems — including soils — I return again and again to one principle:
Diversity matters.
Diversity provides plants with more opportunities to form beneficial symbioses.
Diversity increases resilience to disturbance and stress.
Diversity increases functional redundancy, ensuring that key ecosystem processes continue even when conditions change.
A diverse bacterial community, for example, can contribute a wide range of beneficial functions, including:
nitrogen fixation
phosphorus and iron solubilization
phytohormone and enzyme production
auxin production
biocontrol and disease suppression
bioremediation
plant stress reduction
carbon cycling and sequestration
With this in mind, I returned to my friend’s original question.
Nitrogen-Fixing Bacteria and Rhizobacteria
Nitrogen-fixing bacteria are commonly discussed under the umbrella term rhizobacteria, which broadly refers to bacteria that live in close association with plant roots. One of the most well-known nitrogen-fixing groups within this category is Rhizobium, which belongs to the bacterial order Rhizobiales.
DNA sequencing allows us to detect the presence and relative abundance of Rhizobiales in compost extracts. However, it is important to be precise:
Rhizobacteria is an ecological term, not a taxonomic one.
Rhizobiales is a taxonomic group that includes many nitrogen-fixing species.
The presence of these organisms does not automatically mean nitrogen fixation is occurring in soil or being supplied to plants.
Other bacterial groups also contribute to nitrogen cycling, including members of the Actinobacteria, Bacilli, and Pseudomonadales.
I personally think we should be looking at all of these rhizobacteria when considering Nitrogen-fixing bacteria.
What the DNA Data Show
The Krona plots below illustrate the relative abundance of key plant-associated and nitrogen-fixing bacterial groups detected in my three compost extracts:
Rhizobiales (orange)
Actinobacteria (purple)
Bacilli (blue)
Pseudomonadales and Flavobacteriales (green)
Percent Compostition of Rhizobacteria in Rewild's Compost Extracts
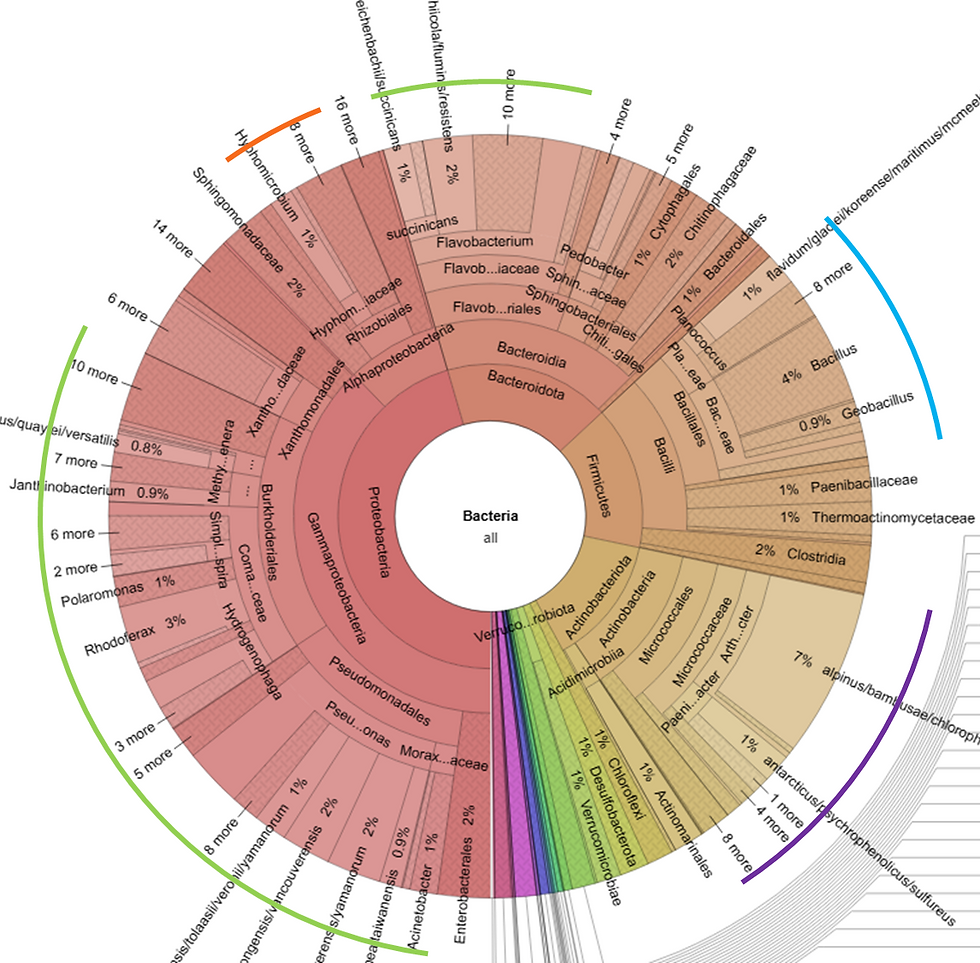
Figure 1. 16S Krona plot of Rewild Early-Ex.

Figure 2. 16S Krona plot of Rewild Mid-Ex.

Figure 3. 16S Krona Plot of Rewild Late-Ex.
In all of these Krona Plots, the composition of N-fixing rhizobacteria makes up approximately 50-60% of the microbial community. All of these extracts can and should be considered rhizobial inoculants.
To simplify interpretation, I also summarized these results in tabular form (Table 1), showing the percent composition of beneficial bacterial groups within each extract’s DNA profile, and incorporated the Shannon Diversity Index.
Table 1. Percent composition of known N-fixing groups in 5 different compost extracts

Sources:
Cornwallis Farms Ltd. Vermicompost Extract data from Dr. Zack Jones https://www.aggregodata.com/post/johnson-su-compost---extract-communities-and-performance-data
Living Roots Compost Tea (Troy Hinke) Vermi-Ex from Dr. Zack Jones https://www.aggregodata.com/post/vermicompost-tea-and-extract-communities-created-by-brewmaster-troy-hinke
Comparing Results with Other Compost Systems
Dr. Zack Jones of Aggrego Data has sequenced more than 200 vermicompost, compost extract, and compost tea samples. His work consistently shows that vermicomposts tend to have high Shannon Diversity Index values, often around 6, with values above 5 considered highly diverse.
Interestingly, Dr. Jones also found that vermicomposts tend to maintain relatively consistent communities of beneficial bacteria despite changes in feedstock.
When I compared my results to his publicly available data — including a Johnson-Su compost extract and a vermicompost extract — I noticed a striking similarity. In my three compost extract blends (Early, Mid, and Late Succession), the relative abundance of Rhizobiales was approximately 4%, 5%, and 5%, respectively. These values closely matched Dr. Jones’ findings for both Johnson-Su and vermicompost extracts, which also showed Rhizobiales at around 5%.
This consistency raises an interesting hypothesis: well-made composts, regardless of production method, may converge on similar relative abundances of certain key plant-associated bacterial groups.
What DNA Percentages Don’t Tell Us
It is essential to emphasize that DNA percent composition is not a measure of biomass. While relative abundance can provide useful context, it does not tell us how many organisms are present or whether they are active.
Bacterial biomass is more directly estimated using:
Colony Forming Units (CFUs), where available
Microscopy-based biomass measurements
Percent DNA composition may loosely correlate with biomass when interpreted alongside these tools, but it should never be treated as a standalone metric.
Microscopy-Based Biomass Context
I have conducted standardized Soil Food Web microbial analyses on the three compost blends used to produce these extracts. While sample sizes are still small and further testing is needed, initial results provide helpful context.

Bacterial biomass differed substantially among compost blends. As expected, the Early-Successional compost had the highest bacterial biomass, while biomass decreased progressively in the Mid- and Late-Successional blends as fungal dominance increased.
According to Dr. Elaine Ingham’s guidelines, bacterial biomass around 1,000 µg/g is suitable for mid- to upper-successional plant systems. The Late-Successional compost, with lower bacterial biomass (~250 µg/g), is still appropriate for all plant systems due to its higher fungal contribution and overall trophic balance.
Practical Takeaways
Taken together, these results suggest that most well-made composts contain substantial communities of beneficial and nitrogen-associated bacteria, including Rhizobiales, often at similar relative abundance.
Other factors to consider when choosing a compost/extract as an inoculum are:
overall microbial biomass
biological diversity (eg. Shannon Diversity Index, presence of other soil food web groups like fungi, protists and nematodes)
handling and storage practices (especially the age of the compost and whether moisture was maintained around 35-45%)
certification requirements
cost and availability
However, when both relative abundance and absolute biomass are considered, compost choice becomes more nuanced.
Based on what I know about my own materials, if my primary goal were to maximize rhizobacterial abundance and bacterial biomass, I would choose my Early-Successional compost blend. This material could be applied:
as a seed inoculant (via soaking and drying)
in-furrow at planting
as a soil drench during the growing season (avoiding drought conditions)
It could also be used as a dry seed dressing with a sticking agent such as yucca extract.
My composts have the added benefit of being locally made and regularly evaluated under the microscope, with biological completeness confirmed at the time of sale.
Closing Thoughts
As with most biological questions, this analysis raises more questions than it answers. If you’re curious — or skeptical — I welcome the conversation.
Yours in soil studies,
Colleen

Comments